REM Behavior Disorder (RBD) as an Early Marker for Development of Neurodegenerative Diseases
Umesh Vyas and Rose Franco
Cite this article as: BJMP 2012;5(1):a506
|
|
Abstract REM behavior disorder (RBD) is a parasomnia characterized by emergence of purposeful complex motor activity with an enactment of dream related activities. This condition is associated with vivid often violent dreams. In normal adults during REM, diffuse hypotonia of muscles occur and on polysomnography the limb and chin electromyographic (EMG) channels demonstrate a low voltage or even flat signal. In RBD, the EMG demonstrating intermittent loss of electromyographic atoniais one of the criteria for diagnosis. Diagnostic polysomnographyrequire capturing the complex dream behaviors on video and electroencephalography monitoring confirms that the behavior originated out of REM sleep. RBD can be either idiopathic or symptomatic of various underlying conditions and may in fact be a prodromal symptom of neurodegenerative disease. It can present acutely which is almost always induced by medications; or develop gradually over months to years. More than half of those with RBD will eventually exhibit signs and symptoms of a degenerative neurologic disorder. A Polysomnogram (PSG) is necessary to diagnose RBD, showing absence of REM sleep atonia and related abnormal behavior. Keywords: REM sleep; REM Behavior Disorder; Neurodegenerative diseases; Parkinson’s disease; Polysomnogram |
Introduction
Normal sleep is divided into Non-REM and REM. REM occurs every 90-120 minutes during adult sleep throughout the night with each period of REM progressing in length such that the REM periods in the early morning hours are the longest and may last from 30-60 minutes. Overall, REM accounts for 20-25% of the sleep time but is weighted toward the second half of the night. During REM sleep with polysomnography monitoring one observes a low voltage mixed frequency amplitude EEG and low voltage EMG in the chin associated with intermittent bursts of rapid eye movements. During the periods of REM breathing becomes irregular, blood pressure rises and the heart rate also increases due to excess adrenergic activity. The brain is highly active during REM and the electrical activity recorded in the brain by EEG during REM sleep is similar to that of wakefulness.
Parasomnias are undesirable, unexpected, abnormal behavioral phenomena that occur during sleep. There are three broad categories in parasomnias. They are
- Disorders of Arousal (from Non-REM sleep)
- Parasomnias usually associated with REM sleep, and
- Other parasomnias which also includes secondary type of parasomnias.
RBD is the only parasomnia which requires polysomnographic testing as part of the essential diagnostic criteria.
Definition of RBD
“RBD is characterized by the intermittent loss of REM sleep electromyographic (EMG) atonia and by the appearance of elaborate motor activity associated with dream mentation” (ICSD-2).1 These motor phenomena may be complex and highly integrated and often are associated with emotionally charged utterances and physically violent or vigorous activities. RBD was first recognized and described by Schenck CH et al. in 1986.2 This diagnosis was first incorporated in the International Classification of Sleep Disorders (ICSD) in 1990. (American Academy of Sleep Medicine)
A defining feature of normal REM sleep is active paralysis of all somatic musculature (sparing the diaphragm to permit ventilation). This result in diffuse hypotonia of the skeletal muscles inhibiting the enactment of dreams associated with REM sleep. In RBD there is an intermittent loss of muscle atonia during REM sleep that can be objectively measured with EMG as intense phasic motor activity (figure 1 and 2).
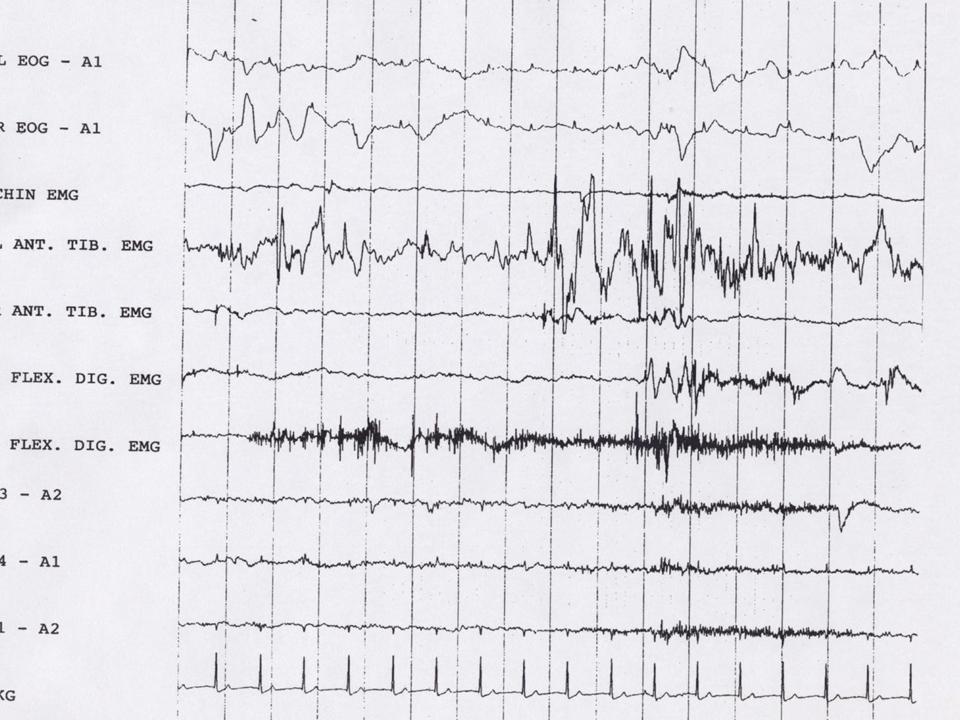
Figure 1
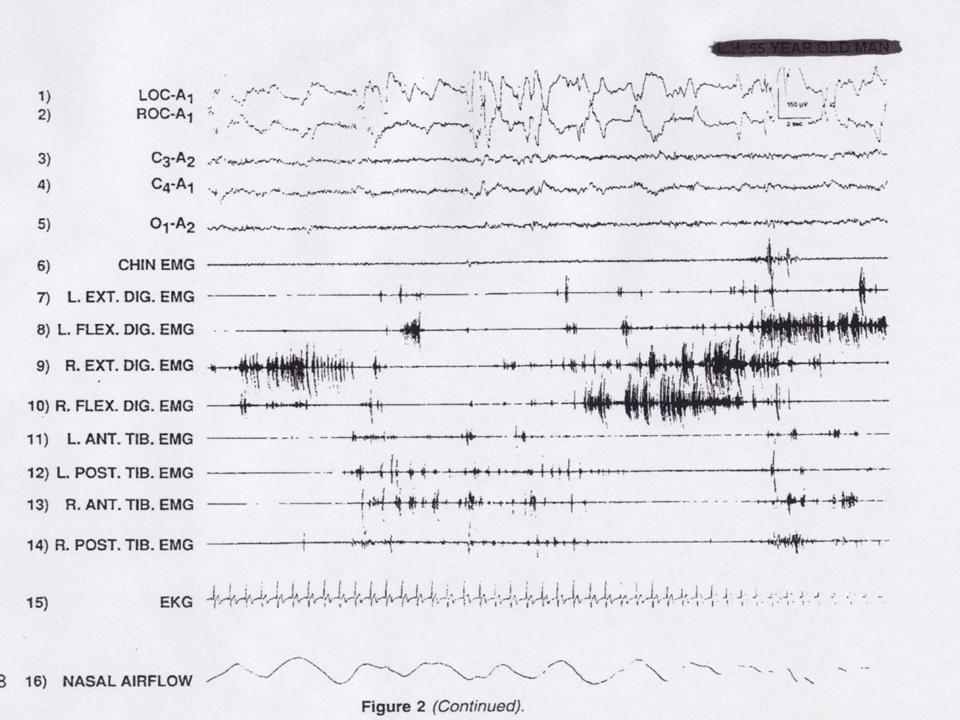
Figure 2
This loss of inhibition often precedes the complex motor behaviors during REM sleep. Additionally, RBD patients will report that their dream content is often very violent or vigorous dream enacting behaviors include talking, yelling, punching, kicking, sitting, jumping from bed, arm flailing and grabbing etc. and most often the sufferer will upon waking from the dream immediately report a clear memory of the dream which coincides very well with the high amplitude violent defensive activity witnessed. This complex motor activity may result in a serious injury to the dreamer or bed partner that then prompts the evaluation.
Prevalence
The Prevalence of RBD is about 0.5% in general population.1, 3 RBD preferentially affect elderly men (in 6th and 7th decade) with ratio of women to men being 1 to 9.4 The mean age of disease onset is 60.9 years and at diagnosis is 64.4 years.5 RBD was reported in an 18 year old female with Juvenile Parkinson disease,6 so age and gender are not absolute criteria.
In Parkinson disease (PD) the reported prevalence ranges from 13-50%,7, 14-19 LewyBody Dementia (DLB) 95%,8 and Multiple System Atrophy (MSA) 90 %.9 The presence of RBD is a major diagnostic criterion for MSA. RBD has been reported in Juvenile Parkinson disease, and pure autonomic failure10-12 all neurodegenerative disorders are synucleinopathies.13
Physiology
The neurons of locus coeruleus, raphe nuclei, tuberomammillary nucleus, pedunculopontine nucleus, laterodorsal tegmental area and the perifornical area are firing at a high rate, and cause arousal by activating the cerebral cortex. During REM sleep, the aforementioned excitatory areas fall silent with the exception of the pedunculopontine nucleus and laterodorsal tegmental areas. These regions project to the thalamus and activate the cortex during REM sleep. This cortical activation is associated with dreaming in REM. Descending excitatory fibers from the pedunculopontine nucleus and laterodorsal tegmental area innervate the medial medulla, which then sends inhibitory projections to motor neurons producing the skeletal muscle atonia of REM sleep.20-21
There are two distinct neural systems which collaborate in the “paralysis” of normal REM sleep, one is mediated through the active inhibition by neurons in the nucleus reticularis magnocellularis in the medulla via the ventrolateral reticulospinal tract synapsing on the spinal motor neurons and the other system suppresses locomotor activity and is located in pontine region.22
Pathophysiology
REM sleep contains two types of variables, tonic (occurring throughout the REM period), and phasic (occurring intermittently during a REM period). Tonic elements include desynchronized EEG and somatic muscle atonia (sparing the diaphragm). Phasic elements include rapid eye movements, middle ear muscle activity and extremity twitches. The tonic electromyogram suppression of REM sleep is the result of active inhibition of motor activity originating in the perilocus coeruleus region and terminating in the anterior horn cells via the medullary reticularis magnocellularis nucleus.
In RBD, the observed motor activity may result from either impairment of tonic REM muscle atonia or from increase phasic locomotor drive during REM sleep. One mechanism by which RBD results is the disruption in neurotransmission in the brainstem, particularly at the level of the pedunculopontine nucleus.23Pathogenetically, reduced striatal dopaminergic mediation has been found24-25 in those with RBD. Neuroimaging studies support dopaminergic abnormalities.
Types of RBD
RBD can be categorized based on severity:
- Mild RBD occurring less than once per month,
- Moderate RBD occurring more than once per month but less than once per week, associated with physical discomfort to the patient or bed partner, and
- Severe RBD occurring more than once per week, associated with physical injury to patient or bed partner.
RBD can be categorized based on duration:
- Acute presenting with one month or less,
- Subacute with more than one month but less than 6 months,
- Chronic with 6 months or more of symptoms prior to presentation.
Acute RBD: In 55 - 60% of patients with RBD the cause is unknown, but in 40 - 45% the RBD is secondary to another condition. Acute onset RBD is almost always induced or exacerbated by medications (especially Tri-Cyclic Antidepressants, Selective Serotonin Reuptake Inhibitors, Mono-Amine Oxidase Inhibitors, Serotonin Norepinephrine Reuptake Inhibitors,26 Mirtazapine, Selegiline, and Biperiden) or during withdrawal of alcohol, barbiturates, benzodiazepine or meprobamate. Selegiline may trigger RBD in patients with Parkinson disease. Cholinergic treatment of Alzheimer’s disease may trigger RBD.
Chronic RBD: The chronic form of RBD was initially thought to be idiopathic; however long term follow up has shown that many eventually exhibit signs and symptoms of a degenerative neurologic disorder. One recent retrospective study of 44 consecutive patients diagnosed with idiopathic RBD demonstrated that 45% (20 patients) subsequently developed a neurodegenerative disorder, most commonly Parkinson disease (PD) or Lewy body dementia, after a mean of 11.5 years from reported symptoms onset and 5.1 years after RBD diagnosis.27
The relationship between RBD and PD is complex and not all persons with RBD develop PD. In one study of 29 men presenting with RBD followed prospectively, the incidence of PD was 38% at 5 years and 65% after 12 years.7, 28, 29 Contrast this with the prevalence of the condition in multiple system atrophy, where RBD is one of the primary symptoms occurring in 90% of cases.9 In cases of RBD, it is absolutely necessary not only to exclude any underlying neurodegenerative disease process but also to monitor for the development of one over time in follow up visits.
Clinical manifestations
Sufferers of RBD usually present to the doctor with complaints of sleep related injury or fear of injury as a result of dramatic violent, potentially dangerous motor activity during sleep. 96% of patients reporting harm to themselves or their bed partner. Behaviors during dreaming described include talking, yelling, swearing, grabbing, punching, kicking, jumping or running out of the bed. One clinical clue of the source of the sleep related injury is the timing of the behaviors. Because RBD occurs during REM sleep, it typically appears at least 90 minutes after falling asleep and is most often noted during the second half of the night when REM sleep is more abundant.
One fourth of subjects who develop RBD have prodromal symptoms several years prior to the diagnosis. These symptoms may consist of twitching during REM sleep but may also include other types of simple motor movements and sleep talking or yelling.30-31 Day time somnolence and fatigue are rare because gross sleep architecture and the sleep-wake cycle remain largely normal.
RBD in other neurological disorders and Narcolepsy:
RBD has also been reported in other neurologic diseases such as Multiple Sclerosis, vascular encephalopathies, ischemic brain stem lesions, brain stem tumors, Guillain-Barre syndrome, mitochondrial encephalopathy, normal pressure hydrocephalus, subdural hemorrhage, and Tourette’s syndrome. In most of these there is likely a lesion affecting the primary regulatory centers for REM atonia.
RBD is particularly frequent in Narcolepsy. One study found 36% pts with Narcolepsy had symptoms suggestive of RBD. Unlike idiopathic RBD, women with narcolepsy are as likely to have RBD as men, and the mean age was found to be 41 years.32 While the mechanism allowing for RBD is not understood in this population, narcolepsy is considered a disorder of REM state disassociation. Cataplexy is paralysis of skeletal muscles in the setting of wakefulness and often is triggered by strong emotions such as humor. In narcoleptics who regularly experienced cataplexy, 68% reported RBD symptoms, compared to 14% of those who never or rarely experienced cataplexy.32-33 There is evidence of a profound loss of hypocretin in the hypothalamus of the narcoleptics with cataplexy and this may be a link that needs further investigation in the understanding of the mechanism of RBD in Narcolepsy with cataplexy. It is prudent to follow Narcoleptics and questioned about symptoms of RBD and treated accordingly, especially those with cataplexy and other associated symptoms.
Diagnostic criteria for REM Behavior Disorder(ICSD-2: ICD-9 code: 327.42)1
A. Presence of REM sleep without Atonia: the EMG finding of excessive amounts of sustained or intermittent elevation of submental EMG tone or excessive phasic submental or (upper or lower) limb EMG twitching (figure 1 and 2).
B. At least one of the following is present:
i. Sleep related injurious, potentially injurious, or disruptive behaviors by history
ii. Abnormal REM sleep behaviors documented during polysomnographic monitoring
C. Absence of EEG epileptiform activity during REM sleep unless RBD can be clearly distinguished from any concurrent REM sleep-related seizure disorder.
D. The sleep disturbance is not better explained by another sleep disorder, medical or neurologic disorder, mental disorder, medication use, or substance use disorder.
Differential diagnosis
Several sleep disorders causing behaviors in sleep can be considered in the differential diagnosis, such as sleep walking (somnambulism), sleep terrors, nocturnal seizures, nightmares, psychogenic dissociative states, post-traumatic stress disorder, nocturnal panic disorder, delirium and malingering. RBD may be triggered by sleep apnea and has been described as triggered by nocturnal gastroesophageal reflux disease.
Evaluation and Diagnosis
- Detailed history of the sleep wake complaints
- Information from a bed partner is most valuable
- Thorough medical, neurological, and psychiatric history and examination
- Screening for alcohol and substance use
- Review of all medications
- PSG (mandatory): The polysomnographic study should be more extensive, with an expanded EEG montage, monitors for movements of all four extremities, continuous technologist observation and continuous video recording with good sound and visual quality to allow capture of any sleep related behaviors
- Multiple Sleep Latency Test (MSLT): Only recommended in the setting of suspected coexisting Narcolepsy
- Brain imaging (CT or MRI) is mandatory if there is suspicion of underlying neurodegenerative disease.
Management
RBD may have legal consequences or can be associated with substantial relationship strain; therefore accurate diagnosis and adequate treatment is important, which includes non-pharmacological and pharmacological management.
Non-pharmacological management: Acute form appears to be self-limited following discontinuation of the offending medication or completion of withdrawal treatment. For chronic forms, protective measures during sleep are warranted to minimize the risks for injury to patient and bed partner. These patients are at fall risk due to physical limitations and use of medications. Protective measure such as removing bed stands, bedposts, low dressers and applying heavy curtains to windows. In extreme cases, placing the mattress on the floor to prevent falls from the bed has been successful.
Pharmacological management: Clonazepam is highly effective in treatment and it is the drug of choice. A very low dose will resolve symptoms in 87 to 90% of patients.4, 5, 7-34 Recommended treatment is 0.5 mg Clonazepam 30 minutes prior to bed time and for more than 90% of patients this dose remains effective without tachyphylaxis. In the setting of breakthrough symptoms the dose can be slowly titrated up to 2.0 mg. The mechanism of action is not well understood but clonazepam appears to decrease REM sleep phasic activity but has no effect on REM sleep atonia.35
Melatonin is also effective and can be used as monotherapy or in conjunction with clonazepam. The suggested dose is 3 to 12 mg at bed time. Pramipexole may also be effective36-38 and suggested for use when clonazepam is contraindicated or ineffective. It is interesting to note that during holidays from the drug, the RBD can take several weeks to recur. Management of patients with concomitant disorder like narcolepsy, depression, dementia, Parkinson disease and Parkinsonism can be very challenging, because medications such as SSRIs, selegiline and cholinergic medications used to treat these disorders, can cause or exacerbate RBD. RBD associated with Narcolepsy, clonazepam is usually added in management and it is fairly effective.
Follow-up
Because RBD may occur in association with neurodegenerative disorder, it is important to consult a neurologist for every patient with RBD as early as possible, especially to diagnose and provide care plan for neurodegenerative disorder, which includes but not limited to early diagnosis and management, regular follow up, optimization of management to provide better quality of life and address medico-legal issues.
Prognosis
In acute and idiopathic chronic RBD, the prognosis with treatment is excellent. In the secondary chronic form, prognosis parallels that of the underlying neurologic disorder. Treatment of RBD should be continued indefinitely, as violent behaviors and nightmares promptly reoccur with discontinuation of medication in almost all patients.
Conclusions
RBD and neurodegenerative diseases are closely interconnected. RBD often antedates the development of a neurodegenerative disorder; diagnosis of idiopathic RBD portends a risk of greater than 45% for future development of a clinically defined neurodegenerative disease. Once identified, close follow-up of patients with idiopathic RBD could enable early detection of neurodegenerative diseases. Treatment for RBD is available and effective for the vast majority of cases.
Key Points
- Early diagnosis of RBD is of paramount importance
- Polysomnogram is an essential diagnostic element
- Effective treatment is available
- Early treatment is essential in preventing injuries to patient and bed partner
- Apparent idiopathic form may precede development of Neurodegenerative disorder by decades
|
Competing Interests None declared Author Details UMESH VYAS, MD, Chair of Department of Psychiatry, Medical Director of Behavioral Health Unit and Medical Director of Sleep Disorders Center, Psychiatrist and Sleep Disorders Specialist, Mayo Clinic Health System, Mankato, MN, USA. ROSE FRANCO, MD Sleep Medicine Fellowship Director and Associate Professor in Medicine and Otolaryngology, Pulmonologist and Sleep Disorders Specialist, Medical College of Wisconsin, Milwaukee, WI, USA. CORRESPONDENCE: UMESH VYAS, MD, Chair of Department of Psychiatry, Medical Director of Behavioral Health Unit and Medical Director of Sleep Disorders Center, Psychiatrist and Sleep Disorders Specialist, Mayo Clinic Health System, Mankato, MN, USA. Email: Vyas.umesh@mayo.edu |
References

The above article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.




